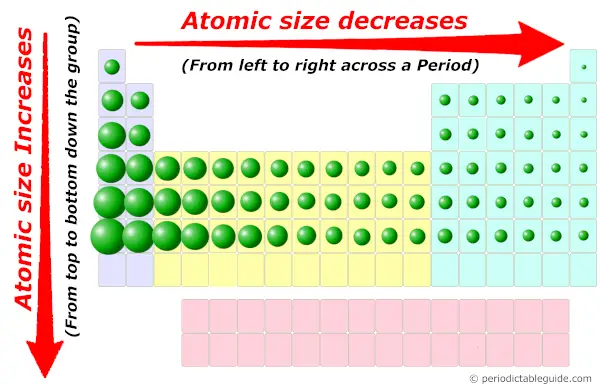
This is an important trend for atomic radius - size decreases left to right, because. First, Mg lies to the left of O on the periodic table. Oxygen is a colourless, odourless reactive gas, the chemical element of atomic number 8 and the life-supporting component of the air. Mg's atomic radius is greater than O's for two reasons. We wish we had saved one of the first edition videos, for the sake of comparison. Oxygen is a chemical element with atomic number 8 which means there are 8 protons and 8 electrons in the atomic structure. 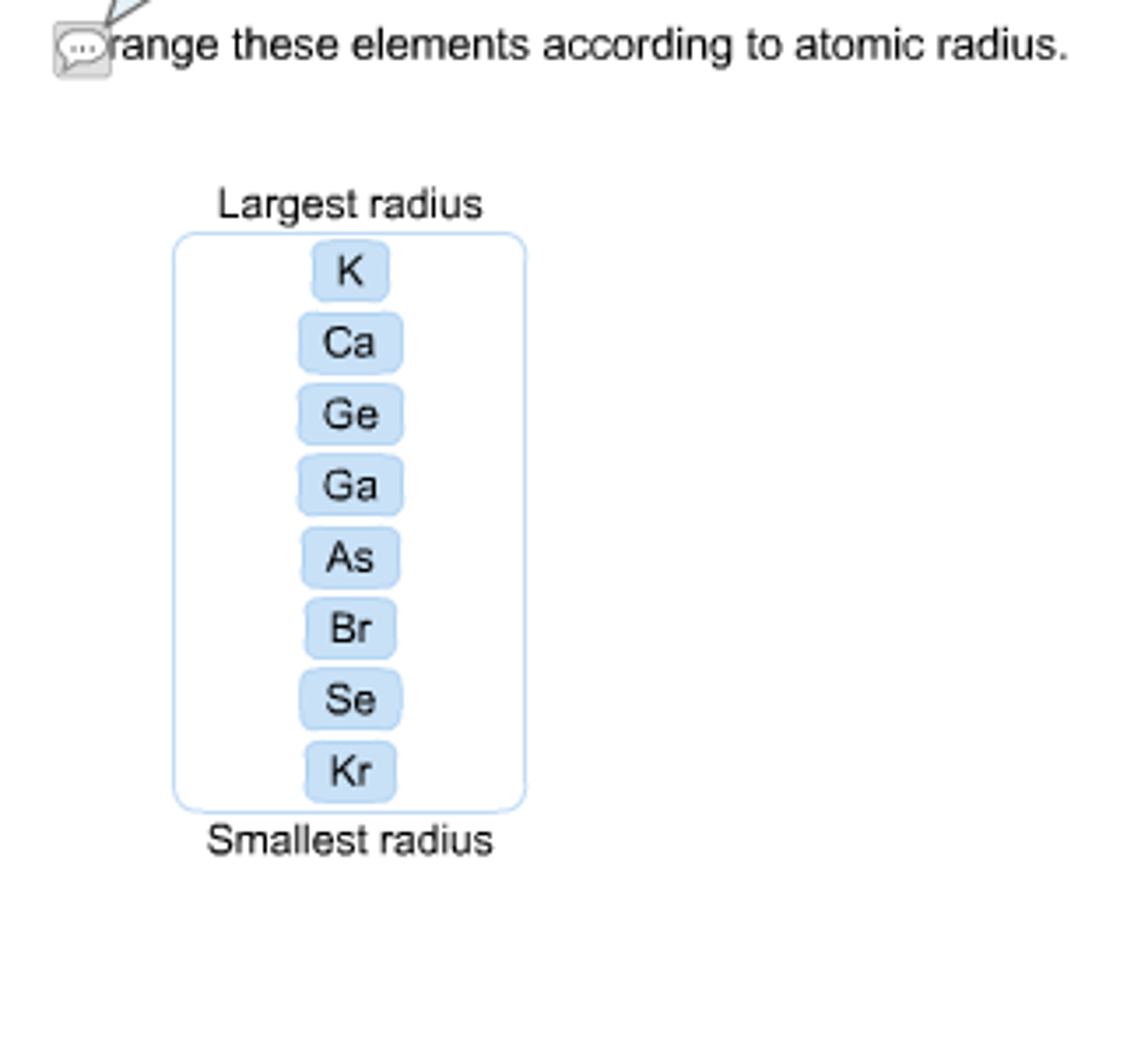
Now the videos stream and the CD seems no longer on offer. Here, zeolites prime concern is to catalytic activity. The animations in the original were huge for the time (hydrogen was about 12 megabytes), so downloading and saving them was a wise use of bandwidth-or one could buy a CD for $37 (20 pounds). Atomic radius is important in the crystal structure prediction. The atomic radius of these elements and ions will increase in the following order: O Al(3+), Si, Al, O(2-) Ag, Rb Cs The semi-colons ( ) indicate a new period (row). From top to bottom in a group, orbitals corresponding to higher. Up to date, curated data provided by Mathematica 's ElementData function from Wolfram Research, Inc. Thus, helium is the smallest element, and francium is the largest. Based on the atomic number of the element, the mass number, and the number of neutrons, three things can be considered. Therefore, the number of neutrons in oxygen (O) is 8. As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. We know that the atomic number of oxygen is 8 and the atomic mass number is about 16. The current version is at least the second. We know that of F 0.72, Further O2- 1.40, O0.73 radii of atoms and ions depends upon relative nuclear charge therefore the radii are in the order. Atomic radii vary in a predictable way across the periodic table. Society of Chemistry (UK) are truly exceptional. What does it mean to measure the "size" of an atom? Elementīoth the content and design of this site created by the Royal
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
